
Barış Genç et al. Clin Transl Med. 2021 Feb.
Background: Upper motor neurons (UMNs) are a key component of motor neuron circuitry. Their degeneration is a hallmark for diseases, such as hereditary spastic paraplegia (HSP), primary lateral sclerosis (PLS), and amyotrophic lateral sclerosis (ALS). Currently there are no preclinical assays investigating cellular responses of UMNs to compound treatment, even for diseases of the UMNs. The basis of UMN vulnerability is not fully understood, and no compound has yet been identified to improve the health of diseased UMNs: two major roadblocks for building effective treatment strategies.
Methods: Novel UMN reporter models, in which UMNs that are diseased because of misfolded superoxide dismutase protein (mSOD1) toxicity and TDP-43 pathology are labeled with eGFP expression, allow direct assessment of UMN response to compound treatment. Electron microscopy reveals very precise aspects of endoplasmic reticulum (ER) and mitochondrial damage. Administration of NU-9, a compound initially identified based on its ability to reduce mSOD1 toxicity, has profound impact on improving the health and stability of UMNs, as identified by detailed cellular and ultrastructural analyses.
Results: Problems with mitochondria and ER are conserved in diseased UMNs among different species. NU-9 has drug-like pharmacokinetic properties. It lacks toxicity and crosses the blood brain barrier. NU-9 improves the structural integrity of mitochondria and ER, reduces levels of mSOD1, stabilizes degenerating UMN apical dendrites, improves motor behavior measured by the hanging wire test, and eliminates ongoing degeneration of UMNs that become diseased both because of mSOD1 toxicity and TDP-43 pathology, two distinct and important overarching causes of motor neuron degeneration.
Conclusions: Mechanism-focused and cell-based drug discovery approaches not only addressed key cellular defects responsible for UMN loss, but also identified NU-9, the first compound to improve the health of diseased UMNs, neurons that degenerate in ALS, HSP, PLS, and ALS/FTLD patients.
Keywords: ALS; HSP; NU-9; PLS; TDP-43 pathology; mSOD1; upper motor neurons.
© 2021 The Authors. Clinical and Translational Medicine published by John Wiley & Sons Australia, Ltd on behalf of Shanghai Institute of Clinical Bioinformatics.
PubMed Disclaimer
The authors declare that there is no conflict of interest.
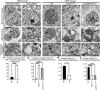 FIGURE 1
FIGURE 1
Upper motor neurons (UMNs) display ultrastructural defects in amyotrophic lateral sclerosis (ALS) patients, and in the mouse models that are diseased due to different underlying causes. (A) Representative electron microscopic (EM) image of UMN of normal control appears intact while (B) UMN of ALS patient showing cytoarchitectural defects. (C) EM image of UMN of WT mouse. (D) Representative EM image of UMN of hSOD1G93A, and (E) prpTDP‐43A315T mouse displaying massive ultrastructural disintegration. (F) The mitochondria in a normal control showing intact inner mitochondrial membranes (arrows), as opposed to (G) mitochondria in ALS patient that displays disintegration of inner mitochondrial membrane (arrowheads). (H) Mitochondria in WT mouse appears to be structurally intact with distinct inner and outer mitochondrial membranes (arrow), whereas (I) mitochondria in UMN in a hSOD1G93A and (J) prpTDP‐43A315T mouse displaying severe disintegration of inner mitochondrial membranes (arrowheads). (K) Electron micrographs of UMN endoplasmic reticulum (ER) in a normal control display properly stacked long cisternae (arrows), but (L) ER in ALS patient shows distension and ballooning of ER cisternae (arrowheads). Similarly, (M) ER in a WT mouse (arrows) looks structurally intact in contrast to (N) ER in UMN of hSOD1G93A and (O) prpTDP‐43A315T mouse that displaying broken, short, and disintegrated ER cisternae (arrowheads). (P) Quantification of average percentage of ER with cytoarchitectural defects/UMN in ALS patients. ****p < .0001, Student's t‐test. (Q) Quantification of average percentage of ER with cytoarchitectural defects/UMN in hSOD1G93A. ****p < .0001, and prpTDP‐43A315T mice. ****p < .0001, One‐way ANOVA followed by Tukey's post hoc multiple‐comparison test. (R) Quantification of average length of ER cisternae/UMN in ALS patients. **p < .001, Student's t‐test. (S) Quantification of average length of ER cisternae/UMN in hSOD1G93A **** p < .0001, and prpTDP‐43A315T mice. ****p < .0001, One‐way ANOVA followed by Tukey's post hoc multiple‐comparison test. Scale bars: A–E = 2 µm; F–O = 200 nm
 FIGURE 2
FIGURE 2
NU‐9 treatment improves ultrastructural integrity of both mitochondria and endoplasmic reticulum (ER) of upper motor neurons (UMNs) that become diseased from mutant SOD1 toxicity. (A) Progression from hit to lead to NU‐9 with chemical structure of NU‐9. (B) Experimental design for in vivo studies. (C–F) Representative electron microscopic (EM) images of UMNs in the motor cortex of WT‐UeGFP mice treated with vehicle at P120. Arrows point to mitochondria with intact inner membrane and ER with intact cisternae. Scale bars, C: 2 µm; D–F: 200 nm. (G–J) Representative EM images of UMNs in the motor cortex of hSOD1G93A‐UeGFP mice treated with vehicle at P120. (G) The cytoplasm is mostly devoid of major key organelles, and (H) there are numerous electron dense aggregates (arrowheads) and large droplets. (I) Mitochondria that lost the integrity of its inner membrane (arrowhead) or have overall structural damage, and (J) fragmented pieces of the ER (arrowheads) are evident. Scale bars, G: 2 µm; H–J: 200 nm. (K–N) Representative EM images of UMNs in the motor cortex of hSOD1G93A‐UeGFP mice treated with 100 mg/kg/day NU‐9. (K) Overall improvement in the cytoplasm with the presence of numerous organelles. (L) Mitochondria appear to be healthy (arrows), and the cytoplasm lacks major dense aggregates and droplets. (M) The integrity of the inner mitochondrial membrane and cristae structure is restored (arrows), and (N) ER cisternae (arrows) are arranged in proper structure without any fragmentation. Scale bars, I: 2 µm; L–N: 200 nm. (O) Quantification of total number of mitochondria/UMN in hSOD1G93A‐UeGFP mice with NU‐9 treatment. **p < .006, One‐way ANOVA followed by Tukey's post hoc multiple‐comparison test. (P) Quantification of percentage of healthy mitochondria/UMN in hSOD1G93A‐UeGFP mice with NU‐9 treatment. ****p < .0001, One‐way ANOVA followed by Tukey's post hoc multiple‐comparison test. (Q) Quantification of the number of ER cisternae/UMN in hSOD1G93A‐UeGFP mice with NU‐9 treatment. *p < .016, One‐way ANOVA followed by Tukey's post hoc multiple‐comparison test. (R) Quantification of average length of ER cisternae/UMN in hSOD1G93A‐UeGFP mice with NU‐9 treatment. **p < .002, One‐way ANOVA followed by Tukey's post hoc multiple‐comparison test
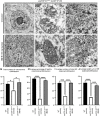 FIGURE 3
FIGURE 3
NU‐9 treatment reduces misfolded SOD1 levels in upper motor neurons (UMNs) of hSOD1G93A‐UeGFP mice. (A) Representative images of UMNs and B8H10 antibody staining that recognizes misfolded SOD1 protein in the motor cortex of WT‐UeGFP or (B) hSOD1G93A‐UeGFP mice treated with vehicle, (C) 20 mg/kg/day NU‐9, or (D) 100 mg/kg/day NU‐9. Scale bars, 20 µm; n ≥ 3 biological replicates. (E) Average integrated density of misfolded SOD1 fluorescence in UMNs in the motor cortex of WT‐UeGFP or hSOD1G93A‐UeGFP mice treated with vehicle, 20 mg/kg/day NU‐9, or 100 mg/kg/day NU‐9; mean, SEM, and individual data points shown for n ≥ 3 biological replicates. **p < .01, ****p < .0001, One‐way ANOVA followed by Tukey's post hoc multiple‐comparison test
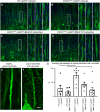 FIGURE 4
FIGURE 4
NU‐9 treatment improves cytoarchitectural integrity of disintegrating apical dendrites of upper motor neurons (UMNs) that become diseased from misfolded SOD1 toxicity. (A) Representative images of UMN apical dendrites in the motor cortex of WT‐UeGFP or (B) hSOD1G93A‐UeGFP mice treated with vehicle, (C) 20 mg/kg/day NU‐9, or (D) 100 mg/kg/day NU‐9. Boxed areas are enlarged to the right and additional examples are supplied. Scale bars: 50 µm (low mag), 10 µm (high mag inset); n ≥ 6 biological replicates. (E) Representative image of a healthy, intact, and (F) a diseased, disintegrating apical dendrite. Scale bars: 5 µm. (G) Average percentage of UMN apical dendrites with vacuoles per section in the motor cortex; mean, SEM, and individual data points shown for n ≥ 6 biological replicates. *p < .05, **p < .01, one‐way ANOVA followed by Tukey's post hoc multiple‐comparison test
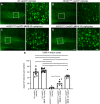 FIGURE 5
FIGURE 5
NU‐9 treatment reduces upper motor neuron (UMN) degeneration of UMNs diseased resulting from misfolded SOD1 toxicity in vivo. (A–D) Representative images of UMNs in the motor cortex of WT‐UeGFP or hSOD1G93A‐UeGFP mice treated with vehicle, 20 mg/kg/day NU‐9, or 100 mg/kg/day NU‐9. Scale bars: 50 µm; n ≥ 5 biological replicates. (E) Average number of UMNs per section in the motor cortex; mean, SEM, and individual data points shown for n ≥ 5 biological replicates. *p < .05, ***p < .001, ****p < .0001, One‐way ANOVA followed by Tukey's post hoc multiple‐comparison test
 FIGURE 6
FIGURE 6
NU‐9 treatment improves ultrastructural integrity of both mitochondria and endoplasmic reticulum (ER) of upper motor neurons (UMNs) that become diseased due to TDP‐43 pathology. Mitochondrial and ER defects in the UMNs of prpTDP‐43A315T‐UeGFP mice were previously published. (A–C) Representative electron microscopic images of UMNs of untreated prpTDP‐43A315T‐UeGFP mice. (A) UMN soma, with few intact organelles. (B) Mitochondria lose integrity of their inner membrane (arrowheads), and (C) ER cisternae are disintegrated and broken (arrowheads). (D–F) Representative electron microscopic images of UMNs of prpTDP‐43A315T‐UeGFP mice treated with 100 mg/kg/day NU‐9. (D) An overall improvement in the cytoarchitecture is evidenced with proper nuclear membrane, presence of numerous healthy organelles, and lack of electron dense aggregates. (E) Mitochondria appear healthy with improved inner membrane and cristae (arrow) (F), and ER cisternae arranged in proper structure with ribosomes attached (arrows). Scale bars: A,D: 2 µm; B,C,E,F: 200 nm. (G) Quantification of the total number of mitochondria/UMN in prpTDP‐43A315T‐UeGFP mice with 100 mg/kg/day NU‐9 treatment; *p < .03. (H) Quantification of percentage of healthy mitochondria/UMN in prpTDP‐43A315T‐UeGFP mice with 100 mg/kg/day NU‐9 treatment; ****p < .0001. (I) Quantification of the number of ER cisternae/UMN in prpTDP‐43A315T‐UeGFP mice with 100 mg/kg/day NU‐9 treatment; ***p < .0003. (J) Quantification of average length of ER cisternae/UMN in prpTDP‐43A315T‐UeGFP mice with 100 mg/kg/day NU‐9 treatment; ****p < .0001. One‐way ANOVA followed by Tukey's post hoc multiple‐comparison test was used for statistical analyses
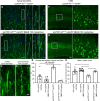 FIGURE 7
FIGURE 7
NU‐9 treatment improves cytoarchitectural integrity of disintegrating apical dendrites and eliminates progressive degeneration of upper motor neurons (UMNs) that become diseased due to TDP‐43 pathology. (A) Representative images of UMN apical dendrites in the motor cortex of untreated prpTDP‐43A315T‐UeGFP mice and (B) prpTDP‐43A315T‐UeGFP mice treated with 100 mg/kg/day of NU‐9. Boxed area is enlarged to the right with additional representative examples. Scale bars: 50 µm (low mag), 10 µm (high mag inset); n ≥ 3 biological replicates. (C) Representative image of a healthy, intact, and (D) a diseased, disintegrating apical dendrite. Scale bars: 5 µm. (E) Average percentage of apical dendrites with vacuoles per section area in the motor cortex; mean, SEM, and individual data points shown for n ≥ 3 biological replicates. **p < .01, ***p < .001, One‐way ANOVA followed by Tukey's post hoc multiple‐comparison test. (F) Representative images of UMNs in the motor cortex of untreated prpTDP‐43A315T‐UeGFP mice and (G) prpTDP‐43A315T treated with 100 mg/kg/day NU‐9. Scale bars: 50 µm; n ≥ 3 biological replicates. (H) Average number of UMNs per section area in the motor cortex; mean, SEM, and individual data points shown for n ≥ 3 biological replicates. *p < .05, **p < .01, ***p < .001, One‐way ANOVA followed by Tukey's post hoc multiple‐comparison test
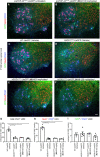 FIGURE 8
FIGURE 8
NU‐9 treatment does not improve lower motor neuron (LMN) degeneration resulting from misfolded SOD1 toxicity in vivo. (A and B) Representative images of LMNs in the lumbar spinal cord of untreated prpTDP‐43A315T‐UeGFP mice or treated with 100 mg/kg/day NU‐9. Scale bar: 50 µm; n ≥ 3 biological replicates. (C–F) Representative images of LMNs in the lumbar spinal cord of WT‐UeGFP or hSOD1G93A‐UeGFP mice treated with vehicle, 20 mg/kg/day NU‐9, or 100 mg/kg/day NU‐9. Scale bar: 50 µm; n = 5 biological replicates. (G) Average number of ChAT+ LMNs per section in the lumbar spinal cord; mean, SEM, and individual data points shown for n = 5 biological replicates. ****p < .0001, One‐way ANOVA followed by Tukey's post hoc multiple‐comparison test. (H) Average number of NeuN+/ChAT+ LMNs (vulnerable to degeneration) per section in the lumbar spinal cord; mean, SEM, and individual data points shown for n = 5 biological replicates. ****p < .0001, One‐way ANOVA followed by Tukey's post hoc multiple‐comparison test. (I) Average number of GFP+/ChAT+ LMNs (resistant to degeneration) per section in the lumbar spinal cord; mean, SEM, and individual data points shown for n = 5 biological replicates
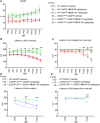 FIGURE 9
FIGURE 9
Behavioral data of WT‐UeGFP, hSOD1G93A‐UeGFP, and prpTDP‐43A315T‐UeGFP mice with or without NU‐9 treatment. WT‐UeGFP and hSOD1G93A‐UeGFP mice were tested every 7 days between postnatal day (P)60 and P120; prpTDP‐43A315T‐UeGFP mice were tested at P60, P90, and P120. (A) Weight of the mice in grams. Mean and SEM shown for n ≥ 5 mice per group. WT‐UeGFP (vehicle) versus hSOD1G93A‐UeGFP (vehicle); # p < .05, two‐way ANOVA with Tukey's multiple‐comparison test. (B) Latency to fall on accelerating rotarod in seconds. Mean and SEM shown for n ≥ 5 mice per group. WT‐UeGFP (vehicle) versus hSOD1G93A‐UeGFP (vehicle). # p < .05, ## p < .01, ### p < .001, #### p < .0001, Two‐way ANOVA with Tukey's multiple‐comparison test. (C) Latency to fall hanging upside down from a wire grid in seconds. Mean and SEM shown for n ≥ 5 mice per group. WT‐UeGFP (vehicle) versus hSOD1G93A‐UeGFP (vehicle). # p < .05, ### p < .001, #### p < .0001; hSOD1G93A‐UeGFP (vehicle) versus hSOD1G93A‐UeGFP (NU‐9 100 mg/kg/day). *p < .05, ***p < .001, ****p < .0001, Two‐way ANOVA with Tukey's multiple‐comparison test. (D) Latency to fall on accelerating rotarod in seconds. Mean and SEM shown for n ≥ 4 mice per group. WT‐UeGFP (vehicle) versus prpTDP‐43A315T‐UeGFP (vehicle). ## p < .01, #### p < .0001, Mixed‐effects analysis with Tukey's multiple‐comparison test. (E) Latency to fall hanging upside down from a wire grid in seconds. Mean and SEM shown for n ≥ 3 mice per group. WT‐UeGFP (vehicle) versus prpTDP‐43A315T‐UeGFP (vehicle), #### p < .0001; prpTDP‐43A315T‐UeGFP (untreated) versus prpTDP‐43A315T‐UeGFP (NU‐9 100 mg/kg/day), ****p < .0001, mixed‐effects analysis with Tukey's multiple‐comparison test